
DNAe. The first true point-of-need Next-Generation Sequencing company.

DNae. The first true point-of-need Next-Generation Sequencing company.
Our mission is to bring dramatic, life-changing improvements to healthcare and beyond with fast, simple and scientifically sound products. To this end, we apply the best scientific skills and energy to build an evolving and innovative suite of electronic microchip-based solutions that delivers faster, simpler and more cost-effective DNA analysis.


Regius Professor Chris Toumazou FRS, FREng, FMedSci founded the company in 2003, as a spin-out from Imperial College London, having invented a way of detecting protons released during DNA synthesis to enable DNA sequence detection using a standard silicon-chip based transistor. This optics-free, label-free method shifted DNA sequencing from specialized, expensive genome centres into the mainstream of local labs and primary care clinics.
Since its foundation, our company has grown to encompass the vast range of disciplines required to build molecular interfaces and assays on a semiconductor platform. Our core competencies in electronic engineering, molecular biology, fluidics and chemistry make us a force to be reckoned with. A force for good.
Headquartered in London, UK, and operating from Washington, DC, our company expanded operations further in 2015 with a clinical diagnostic development site in San Diego, CA.
DNAe is committed to consistently providing high quality products and services for its customers and meeting regulatory requirements through adherence to its established Quality Management System and a commitment to maintaining its effectiveness.
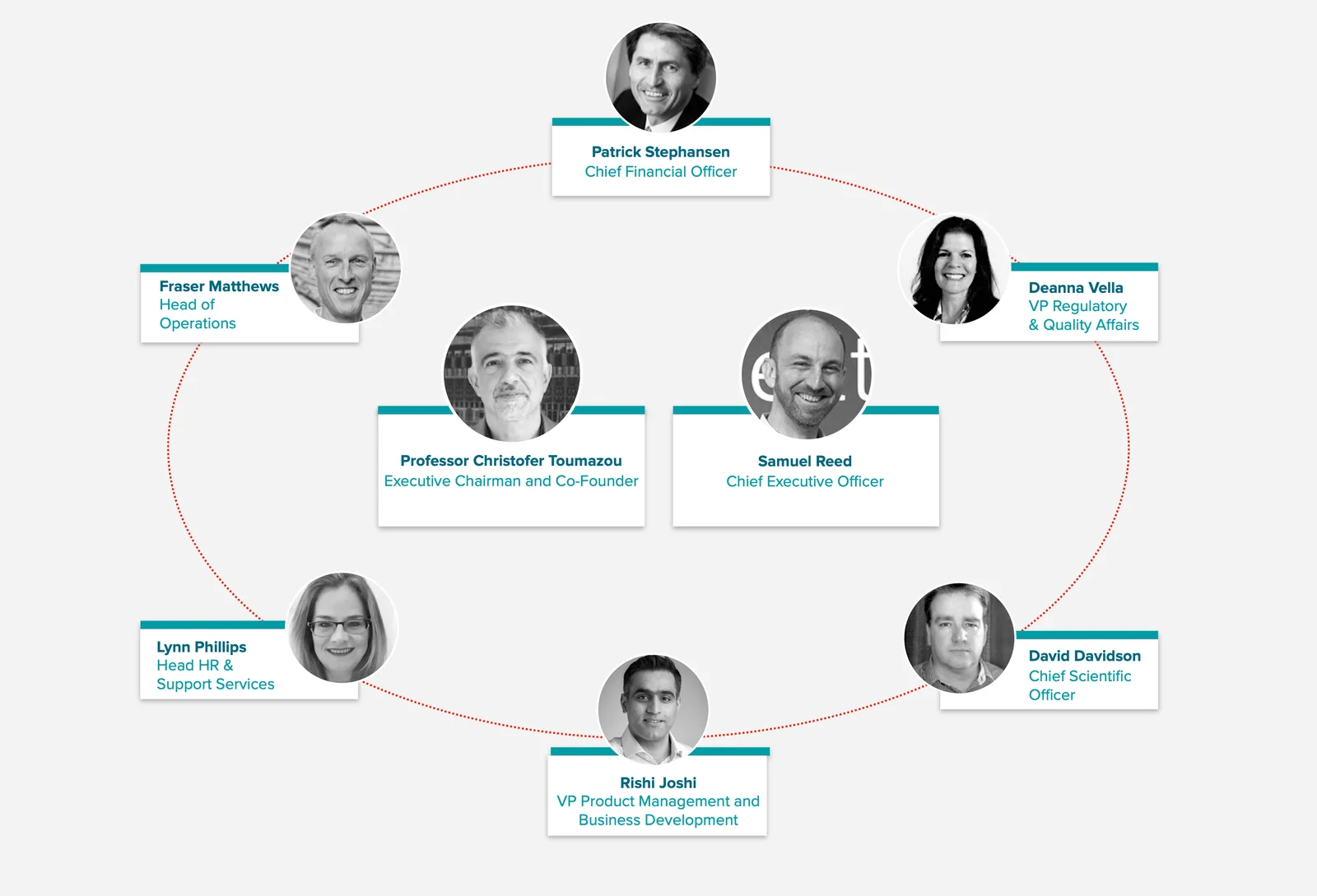
Senior Management Team
Board of Directors
Samuel Reed

An experienced leader of organizations and teams that are commercializing new products based on innovative technologies. Eighteen years of leadership and management experience – majority of which in medical diagnostics, point-of-care, or sequencing. From early research through FDA clearance and market success. Has led operations as well as supporting external clients – from blue chips to start ups.
Tan Sri Lim Kok Thay

Director Tan Sri Lim Kok Thay is the Chairman and Chief Executive of Genting Berhad, the holding company of the Genting Group. The Genting Group is one of Asia’s most dynamic conglomerates and is involved in leisure and hospitality, power generation, oil palm plantations, property development, biotechnology and oil and gas business activities. These business activities are undertaken by the Group’s key companies, namely Genting Malaysia Berhad, Genting Singapore Plc, Genting Hong Kong Limited, Genting Plantations Berhad and the Genting Energy division.
Professor Christofer Toumazou

Tan Kong Han

The Genting Group comprises Genting Berhad (KLSE: Genting) and its listed companies Genting Malaysia Berhad (KLSE: GENM), Genting Plantations Berhad (KLSE: GENP) and Genting Singapore Limited (SGX: G13), as well as principal unlisted subsidiaries Genting Energy Limited and Resorts World Las Vegas LLC.
Tan is also the Chief Executive and Executive Director of Genting Plantations Berhad and oversees the Energy Division of the Genting Group.
Prior to joining Genting Berhad in 2007, Tan worked as an investment banker for more than 13 years and in a Malaysian power generation, entertainment and real estate conglomerate from 2003 to 2007. He is a qualified Barrister at Law, Lincoln’s Inn. He holds a MA from the University of Cambridge.
Advisory Board

Caroline Popper, M.D MPH J.D.
Associate Professor of Medicine, Associate Epidemiologist, Senior Consultant in Infectious Diseases, Infection Control Programme Geneva University Hospitals and Faculty of Medicine, Geneva, CH
Stephan Harbarth earned his medical degree from Ludwig-Maximilians-University in Munich in 1993 and completed his residency in internal medicine and tropical medicine at Munich University Hospitals. After serving as a clinical fellow in the Infectious Diseases Division and Infection Control Program in the Department of Internal Medicine at Geneva University Hospitals, Dr Harbarth completed his master’s degree in epidemiology at Harvard University in Boston. He is board certified in infectious diseases and was appointed associate professor at the University of Geneva in 2010. Dr Harbarth’s work has garnered several awards, including the ICAAC/ASM Young Investigator Award (2003), the Young Investigator Award from ESCMID (2006), the Swiss Society for Infectious Diseases Award for epidemiological research (2008, 2011), and the SHEA Investigator Award (2011). Dr Harbarth’s group is currently conducting several clinical and epidemiological studies to evaluate key questions related to the control of the acquisition, transmission and infection by multidrug-resistant microorganisms. They participate in several ongoing large-scale EU-funded studies to address this public health threat and collaborate closely with the Genomics Research Laboratory at HUG, based on a productive translational research platform.

Nick McCooke
Founding CEO of Solexa
Nick is one of the pioneers of Next Generation Sequencing (NGS). He was the founding CEO of Solexa, building and leading the Cambridge-based team that developed the technology that would become the Illumina sequencing platform. Critically, he brought together the Cambridge chemistry with the Swiss array technology to forge the successful platform that remains the core of the Illumina product line today.
He is an advocate of ‘imagining the future’. In 2001, he coined the phrase the ‘$1,000 genome’ to drive awareness of the vast potential of NGS. In an interview with the BBC in 2002, he predicted a time when an individual’s genome sequence would form part of their medical records.
Nick remains involved with DNA-related technologies as non-executive director, consultant and mentor. He co-founded and was CEO of a Sydney based DNA sequencing company, which he successfully exited in 2021. Nick is an engineer by academic background and has an MBA from the London Business School.

Nathan Ledeboer, PhD
Professor of Pathology, Chief of Clinical Pathology at Medical College of Wisconsin
Nathan A. Ledeboer is a Professor and Chief of Clinical Pathology in the Department of Pathology and Laboratory Medicine at the Medical College of Wisconsin and the Associate Chief Medical Laboratory Officer for Froedtert Health in Milwaukee, WI. His research endeavors, particularly in the area developing diagnostic tools for infectious diseases, have led to numerous publications in peer-reviewed journals and more than 200 funded research projects.
